Aminoglycoside Nephrotoxicity Risk Estimator
Select the patient characteristics below to estimate the potential risk level for kidney damage during aminoglycoside therapy.
Low
It is a frustrating irony of modern medicine: some of our most powerful weapons against life-threatening bacterial infections can simultaneously attack the very organs tasked with clearing them from the body. Aminoglycoside nephrotoxicity is a dose-dependent form of kidney damage caused by the accumulation of aminoglycoside antibiotics in the proximal tubules of the renal cortex. While these drugs are lifesavers for severe Gram-negative infections, they carry a heavy price tag for the kidneys, with 10-25% of patients experiencing some level of renal impairment during treatment.
The Heavy Hitters: What are Aminoglycosides?
When doctors face a multidrug-resistant infection, they often turn to the aminoglycoside class. These are bactericidal agents, meaning they don't just stop bacteria from growing-they kill them. The most common examples include Gentamicin, an antibiotic widely used for sepsis and complicated urinary tract infections, Tobramycin, and Amikacin. These drugs are essential because they tackle pathogens that other antibiotics simply can't touch. However, they have a natural affinity for kidney tissue, making the renal system a primary target for toxicity.
How the Damage Actually Happens
The process starts after the drug is filtered by the glomeruli. About 5% of the dose doesn't just pass through into the urine; instead, it gets trapped in the epithelial cells of the S1 and S2 segments of the proximal tubules. Think of it as a cellular "clog." The drugs accumulate in endosomal and lysosomal vacuoles and the Golgi complex, triggering a cascade of cellular stress.
For years, we thought this was just a matter of the tubules getting blocked. But modern research, including a major review by Lopez-Novoa in 2011, shows it is far more complex. The damage isn't just structural; it's vascular. The drugs cause renal vasoconstriction-essentially narrowing the blood vessels in the kidney-and mesangial contraction. This combination of cellular death in the tubules and reduced blood flow leads to a drop in the glomerular filtration rate (GFR), causing the kidneys to stop cleaning the blood effectively.
Spotting the Warning Signs
Unlike some types of kidney failure where the patient stops producing urine (oliguria), aminoglycoside-induced damage is usually nonoliguric. This means the patient is still peeing, often more than 400 mL a day, which can dangerously trick clinicians into thinking the kidneys are fine. The real warning signs are hidden in the blood and urine chemistry.
The first red flags aren't usually the creatinine levels, but the "leakage" of electrolytes and proteins into the urine. You'll see an increase in urinary excretion of sodium, potassium, magnesium, and calcium, along with proteins like beta-2-microglobulin. By the time serum creatinine rises by 0.5 mg/dL or jumps 50% above the patient's baseline, the damage is already well underway. Typically, this clinical decline manifests 5 to 7 days after therapy begins.
| Drug Entity | Toxicity Potential | Primary Use Case | Recovery Profile |
|---|---|---|---|
| Gentamicin | High | Broad Gram-negative/Sepsis | Partial to Full (1-3 weeks) |
| Amikacin | Moderate | Multidrug-resistant strains | Partial to Full (1-3 weeks) |
| Tobramycin | Moderate-High | Pseudomonas infections | Partial to Full (1-3 weeks) |
Who is Most at Risk?
Not everyone reacts the same way to these drugs. Certain factors act as "force multipliers" for kidney damage. Age is a big one-patients over 65 are significantly more vulnerable. Pre-existing kidney issues are another critical risk; if a patient's baseline GFR is below 60 mL/min/1.73m², the risk of toxicity jumps by over 3 times.
Then there is the danger of "drug cocktails." Using aminoglycosides alongside Vancomycin, a glycopeptide antibiotic often used for MRSA, increases the risk of acute kidney injury by nearly 2.7-fold. Dehydration also plays a role, as volume depletion concentrates the drug in the renal tubules, making the toxic effect more intense.
Dosing Strategies: Timing Matters
One of the most interesting discoveries in renal pharmacology is that *how* you give the drug is just as important as *how much* you give. Old-school thrice-daily dosing is now known to be far more toxic than once-daily dosing. Why? Because once-daily dosing allows for a "washout period" where the kidney cells can clear the drug before the next dose arrives.
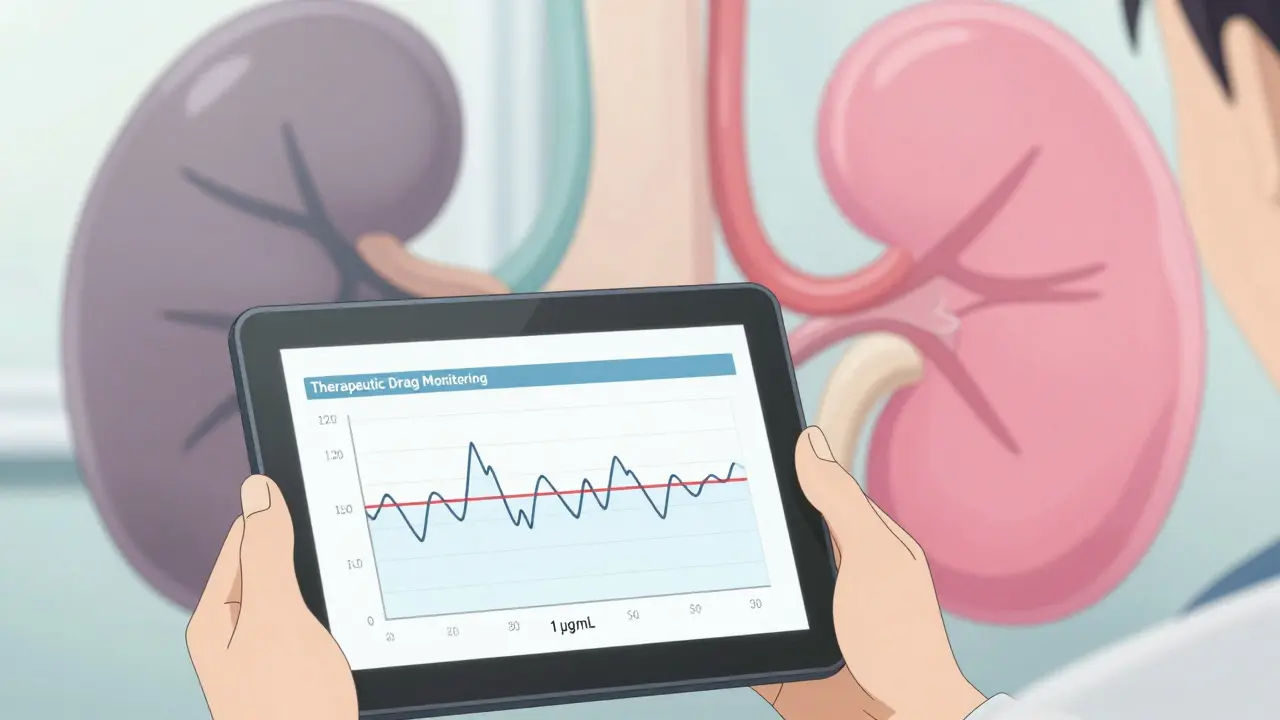
Even the time of day matters. Data suggests that administering the dose around 1:30 p.m. is associated with the lowest rates of nephrotoxicity, pointing to a circadian rhythm in how our kidneys handle toxins. By keeping trough levels-the lowest concentration of the drug in the blood before the next dose-below 1 μg/mL, doctors can significantly reduce the chance of permanent damage.
Can We Prevent the Damage?
While we don't have a magic pill to stop nephrotoxicity yet, research into Polyaspartic acid is promising. This compound acts like a shield, preventing aminoglycosides from binding to the negatively charged phospholipid bilayers of kidney membranes. By blocking this binding, it stops the drug from interfering with lysosomal activities and prevents the formation of "myeloid bodies" (the cellular debris seen under a microscope during kidney injury).
In the real world, the best prevention is rigorous Therapeutic Drug Monitoring (TDM), the clinical practice of measuring drug serum concentrations to optimize dosing. Checking serum creatinine every 48 to 72 hours and adjusting doses based on the patient's real-time renal clearance is the gold standard for keeping patients safe.
The Path to Recovery
The good news is that the kidneys are remarkably resilient. For most people, the recovery process begins 3 to 5 days after the antibiotic is stopped. Complete functional recovery usually takes between one and three weeks. In some cases, a "refractory state" develops where the kidneys actually become less susceptible to the drug after an initial bout of damage, possibly because the regenerating cells are less likely to accumulate the antibiotic.
However, the goal is always to avoid the injury entirely. A Mayo Clinic study showed that while 82% of patients recovered within 30 days, a significant portion still suffered a permanent reduction in renal function. When you're dealing with a drug that can cause acute tubular necrosis, "mostly recovered" isn't always good enough.
How long does it take for aminoglycoside kidney damage to appear?
Typically, nephrotoxicity develops after 5 to 7 days of continuous therapy. It rarely happens overnight; instead, it manifests as a slow rise in serum creatinine and the early appearance of proteins and electrolytes in the urine.
Can kidney damage from Gentamicin be reversed?
Yes, in many cases. Most patients see the start of recovery 3-5 days after stopping the medication, with full recovery occurring within 1-3 weeks. However, some patients may experience a permanent decrease in their overall kidney function depending on the severity of the injury.
Why is once-daily dosing safer than multiple daily doses?
Once-daily dosing allows the drug concentration in the kidney tubules to drop significantly between doses. This "washout period" reduces the total accumulation of the drug in the proximal tubule cells, thereby lowering the risk of cellular toxicity and renal failure.
What is the role of Vancomycin in increasing kidney risk?
Vancomycin is also nephrotoxic. When used together with aminoglycosides, they create a synergistic toxic effect. Meta-analyses suggest that this combination increases the risk of acute kidney injury by approximately 2.7-fold compared to using an aminoglycoside alone.
What are the earliest biomarkers of aminoglycoside nephrotoxicity?
The earliest signs are not blood-based but urine-based. An increase in the excretion of electrolytes (Na+, K+, Mg2+, Ca2+), enzymes like N-acetylglucosaminidase, and proteins such as beta-2-microglobulin often precedes the rise in serum creatinine.
Del Bourne
April 7, 2026 AT 01:55It is so crucial to emphasize the nonoliguric nature of this damage. Many people assume that as long as they are producing urine, their kidneys are functioning normally, but as mentioned, the biochemical markers are the real indicators here. Monitoring serum creatinine is a start, but watching for that electrolyte leakage in the urine is where the real early detection happens!
Benjamin cusden
April 8, 2026 AT 08:08The mention of the 1:30 p.m. dosing window is a quaint touch, though anyone with a basic grasp of chronopharmacology knows that circadian rhythms vary wildly between individual patients. Relying on a generic time of day is practically anecdotal compared to the rigorous application of TDM.
Srikanth Makineni
April 9, 2026 AT 09:25tdm is the only way
Victoria Gregory
April 10, 2026 AT 14:31Wow... it's just so wild how the body works!!! 🌸 I love that the kidneys can be so resilient and start healing almost immediately after the meds stop... nature is truly amazing!!!! ✨🙏
jack hunter
April 11, 2026 AT 00:14its all just a way to push more expensive monitoring tests. why do we act like the 'washout period' isnt just a lucky guess. medicine is basically just trial and eror until someone dies and they call it a side effect.
Ethan Davis
April 11, 2026 AT 11:19Funny how they mention polyaspartic acid as a 'shield' but it's not widely used yet. Probably because the pharmaceutical companies make way more money treating chronic kidney failure than they would from a preventative shield. Follow the money!
Kathleen Painter
April 12, 2026 AT 09:14I think it's really important for us to consider the holistic impact on these patients, especially the elderly, because when you're over 65 and facing a severe infection, the stress on the body is already immense, and adding a nephrotoxic drug like Gentamicin into the mix creates such a fragile balance where the medical team has to be incredibly vigilant about every single milliliter of fluid the patient is taking in to avoid that dangerous concentration in the tubules that leads to permanent damage.
dwight koyner
April 12, 2026 AT 13:54The synergy between Vancomycin and aminoglycosides is a particularly concerning aspect of critical care. When treating MRSA and Gram-negative infections simultaneously, the risk of acute kidney injury becomes a primary concern for the clinical team. It is imperative that we prioritize the lowest effective dose and maintain aggressive hydration to mitigate these risks.
Laurie Iten
April 13, 2026 AT 22:07the irony of saving the life only to damage the filter that keeps the life going is a strange cycle
Jamar Taylor
April 14, 2026 AT 21:08Keep pushing for those better dosing strategies! It's awesome to see the science evolving toward once-daily dosing to give those kidneys a break. We can definitely improve patient outcomes with these insights!
Nikhil Bhatia
April 15, 2026 AT 23:08Too much medical jargon but basically just dont overdose on antibiotics.
Stephen Luce
April 16, 2026 AT 10:30It's honestly scary to think that you can still be peeing normally while your kidneys are actually failing. I've heard a few stories from friends who went through this and they were totally blindsided because they didn't have that typical 'stop peeing' symptom people talk about.