Ever wondered why some medications cost a fortune while others are affordable and do the exact same thing? The secret lies in the complex machinery of the FDA Office of Generic Drugs. This isn't just another government department; it is the gatekeeper that ensures the cheaper version of your prescription is just as safe and effective as the brand-name original. If this office didn't do its job, the balance between drug affordability and patient safety would collapse.
What Exactly Does the OGD Do?
At its core, the Office of Generic Drugs (OGD) is a specialized division within the Center for Drug Evaluation and Research (CDER). Its primary mission is to make sure high-quality, affordable generic drugs are available to the public. To do this, they oversee the development and implementation of strict standards for safety and effectiveness.
The OGD doesn't just check boxes; they manage the entire lifecycle of a generic drug's entry into the US market. This includes reviewing Abbreviated New Drug Applications (ANDAs), which are the specific applications generic manufacturers submit to prove their product is equivalent to the brand-name drug. They ensure the generic version has the same active ingredient, strength, dosage form, and route of administration. If a drug is labeled as a generic, the OGD is the entity that verified it actually works the same way in your body.
The Power Structure: How OGD is Organized
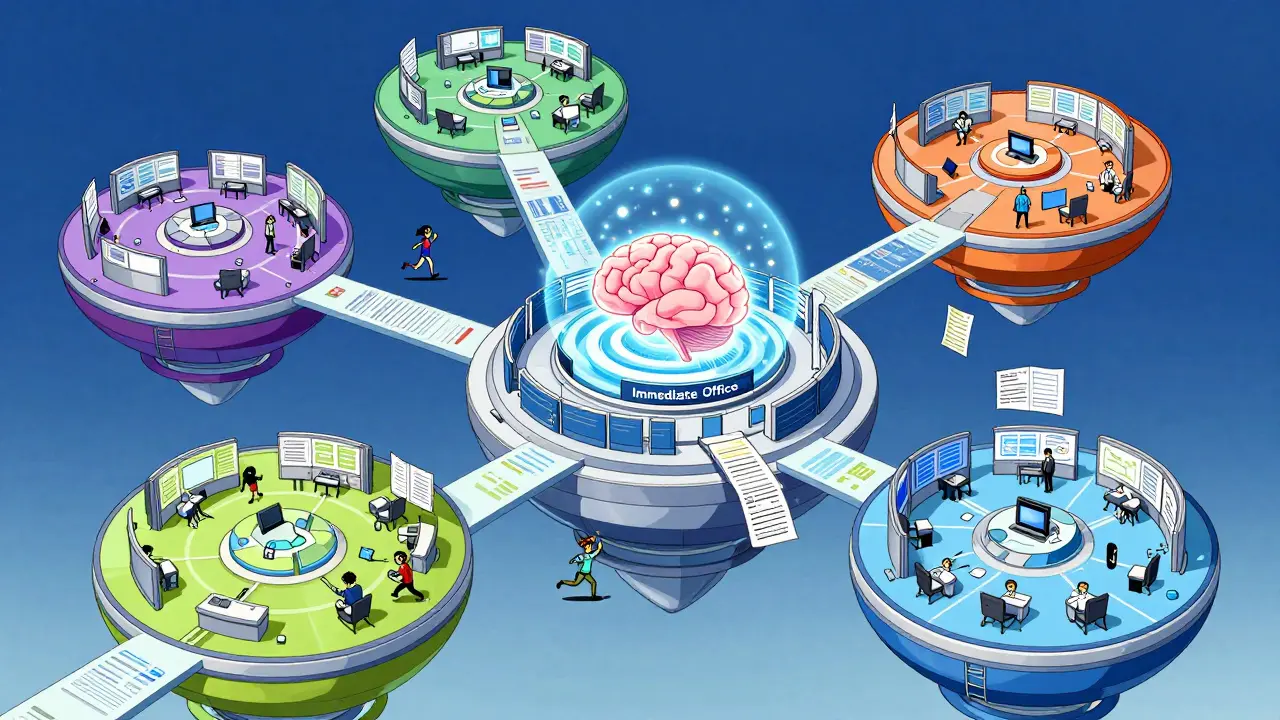
Back in 2013, the FDA realized the generic drug review process was too slow and disjointed. They reorganized the OGD into a "super office" that reports directly to the CDER director. This change gave the office more autonomy and a centralized administrative engine to speed up approvals. Today, the office is split into an Immediate Office and five specialized sub-offices, each with a distinct job.
The Immediate Office (IO) acts as the brain of the operation. It provides the strategic direction and leadership for everything else. Inside the IO, you'll find the Global Generic Drug Affairs Team, which handles international relations, and the Division of Legal and Regulatory Support, which deals with the heavy legal lifting regarding patents.
The five sub-offices handle the technical and operational work:
- Office of Bioequivalence: This team focuses on whether the drug reaches the bloodstream at the right rate and extent. They also run the Safety and Surveillance Team to monitor drugs after they hit the market.
- Office of Generic Drug Policy: These are the rule-makers. They handle the complex legalities of the Hatch-Waxman Act and decide how regulatory policies should evolve.
- Office of Regulatory Operations: This is the engine room. They house the Regulatory Project Managers (RPMs) who keep the ANDA reviews on schedule so the agency meets its goal dates.
- Office of Research and Standards: This group looks at the science. They use quantitative methods and modeling to set the benchmarks for how generic drugs should be tested.
- Office of Safety and Clinical Evaluation: They ensure that the clinical side of the drug-how it affects the patient-is thoroughly vetted.
| Sub-Office | Primary Responsibility | Key Deliverable |
|---|---|---|
| Bioequivalence | Biological performance testing | Bioequivalence specifications |
| Generic Drug Policy | Legal and regulatory frameworks | Hatch-Waxman interpretations |
| Regulatory Operations | Project management and filing | ANDA review timelines |
| Research and Standards | Scientific methodology | Regulatory science plans |
| Safety and Clinical Evaluation | Clinical safety monitoring | Safety labeling and risk alerts |
The Legal Backbone: Hatch-Waxman and GDUFA
You can't talk about the OGD without mentioning the Hatch-Waxman Act. Passed in 1984, this law created the modern generic drug industry. It allows generic companies to skip the massive, expensive clinical trials that brand-name drugs undergo, provided they can prove bioequivalence. The OGD spends a huge amount of its time managing the tension between brand-name patents and the drive for generic competition.
But how does the OGD afford to do all this review work? That's where GDUFA (Generic Drug User Fee Amendments) comes in. Instead of relying solely on taxpayer money, the FDA charges generic drug manufacturers fees to have their applications reviewed. This funding allows the OGD to hire more staff and implement better technology to clear the backlog of applications. It's essentially a "pay-for-performance" model where the industry funds the very regulators that keep them in check.
Key Responsibilities and Daily Operations
If you looked at the OGD's to-do list, it would be a mix of high-level science and intense project management. One of their most critical jobs is managing drug shortages. When a vital medication disappears from pharmacy shelves, the OGD works to expedite the review of "first generics" to bring new competitors into the market quickly.
They also handle Risk Evaluation and Mitigation Strategies (REMS). If a brand-name drug has a strict safety program (like requiring a patient to have a blood test before every refill), the OGD ensures the generic version follows the same safety protocols. This prevents a "safety gap" where a cheaper drug might be distributed without the necessary warnings or restrictions.
Furthermore, the OGD acts as a global leader. Through the Global Generic Drug Affairs Team, they coordinate with other countries' health authorities. Why? Because drug manufacturing is global. A facility in India or China might supply generics for both the US and Europe. By aligning standards internationally, the OGD makes the entire supply chain more stable.
Common Pitfalls in Generic Drug Approval
Getting a drug through the OGD isn't as simple as copying a formula. Many companies fail because they ignore the nuances of the ANDA process. A common mistake is submitting a filing that doesn't perfectly align with the current OGD standards for bioequivalence. If the data is slightly off, the Regulatory Project Manager will flag it, leading to costly delays.
Another hurdle is the legal battle over exclusivity. The Division of Legal and Regulatory Support carefully tracks which brand drugs have "exclusivity periods" where no generics are allowed. If a company tries to jump the gun on a patent certification, they can find themselves in a legal nightmare that lasts years.
What is the difference between an NDA and an ANDA?
A New Drug Application (NDA) is for a brand-new drug and requires full clinical trials to prove safety and efficacy. An Abbreviated New Drug Application (ANDA) is for a generic drug; it "abbreviates" the process by only requiring proof that the generic is bioequivalent to the already-approved brand-name drug.
How does GDUFA affect the cost of generic drugs?
GDUFA requires manufacturers to pay fees to the FDA. While this is a cost to the company, it speeds up the approval process. Faster approvals lead to more generic competitors in the market, which typically drives prices down for the consumer.
Who is responsible for monitoring a generic drug after it is approved?
The OGD Safety and Surveillance Team, located within the Office of Bioequivalence, is responsible for monitoring adverse event reports and ensuring the drug remains safe for public use after it hits the market.
What happens if a generic drug fails a bioequivalence test?
If a drug isn't bioequivalent, the FDA will not approve the ANDA. The manufacturer must go back to the lab, adjust the formulation, or improve the manufacturing process and resubmit their data for review.
Does the OGD handle patents?
The OGD does not grant patents, but its Division of Legal and Regulatory Support makes critical decisions on patent certifications and exclusivity. They determine if a generic can enter the market without infringing on a brand's legal protections.
Next Steps for Industry Stakeholders
If you are a manufacturer looking to navigate the OGD, your first priority should be the GDUFA Regulatory Science Plan. This document tells you exactly where the FDA's priorities lie for the next few years. Aligning your research with these goals can significantly smooth out the review process.
For those dealing with drug shortages, keep a close eye on the OGD's priority submission lists. The agency often fast-tracks reviews for drugs that are in critical shortage, meaning your application could move to the front of the line if you're filling a desperate public health need.

Colleen Tankard
April 18, 2026 AT 05:15It's honestly wild how much work goes into making sure our generic meds aren't just random knock-offs 💊✨. Really makes you appreciate the boring side of government that actually keeps us safe! 💖
Sophia Rice
April 19, 2026 AT 22:39Im glad thier is a system for this. I always worrit about if my generic stuff was actually the same as the brand name one but now i feel way better about it 😊
Ben Ferguson
April 21, 2026 AT 05:30The sheer audacity of the pharmaceutical industry to hold onto patents for decades is absolutely staggering, and while the OGD is doing their best to navigate the Hatch-Waxman Act, it often feels like a Herculean task to actually get a competitive price point for the average consumer who is just trying to survive the month without going bankrupt from a single prescription bottle! It's a dramatic tug-of-war between corporate greed and public health, and we are all just caught in the middle of this regulatory storm, hoping the 'super office' can actually speed things up before we all lose our minds over the cost of basic healthcare in this country!
Rock Stone
April 22, 2026 AT 14:36That's a great perspective on the regulatory side of things. Everyone just needs to stay positive and keep pushing for those affordable options. ✌️
Michael Lewis
April 24, 2026 AT 08:25The GDUFA model is the only way this works. If we don't make the companies pay for the review, the backlog just grows. We need efficiency, and that means funding the experts who can actually spot a bad formulation before it hits the pharmacy shelf. No excuses for slow approvals when the money is there!
Theresa Griffin MEP
April 25, 2026 AT 11:00The regulatory science plan is paramount. Compliance is non-negotiable. Efficiency ensures accessibility.
Anna BB
April 26, 2026 AT 16:34It's interesting to think about the balance between innovation and affordability... like, we need the brand names to invent things, but we need the generics to make them reachable!!! It's a delicate dance of ethics and law... really makes you ponder the value of a human life versus a patent!!!
Heer Malhotra
April 27, 2026 AT 01:27It is an absolute travesty that the world relies so heavily on the US FDA's internal politics. India produces a massive volume of the world's generics, and our manufacturing standards are world-class. The OGD's coordination with international authorities is not a favor they do for us; it is a necessity because without Indian pharmaceutical prowess, the global healthcare system would be in a state of complete collapse. The West must acknowledge the superior efficiency of Eastern production models.
Josephine Wyburn
April 27, 2026 AT 13:16I literally cannot even deal with the stress of trying to find a drug that isn't on shortage right now, and reading about the 'super office' just makes me feel like everything is just a giant bureaucratic nightmare 🙄💅. Like, why is it so hard for them to just make sure there's enough medicine for everyone without it being a whole 'legal nightmare' involving patents and exclusivity periods that basically feel like they're designed to keep us suffering while they play corporate games in their fancy offices 😩😭💔. My anxiety is peaking just thinking about my pharmacy telling me 'sorry we're out' for the third time this month, and honestly, I feel like the whole system is just a joke at this point, just a massive, expensive joke that we all have to pay for with our health and our sanity! 💅✨
Bonnie Piersall
April 28, 2026 AT 10:07That's a real rollercoaster of a process for those manufacturers. One wrong move on a bioequivalence test and boom, you're back to square one in the lab. It's a high-stakes game of scientific leapfrog.